Our QA services truly cater to any size of businesses .MSDS authoring services start from as low as 10$ . Prices may vary depending on the intensity of the technical work involved.Whatever may be the case , contact us for all your laboratory document needs.
We also offer to author your Technical Data Sheets, QA procedures (SOPs), Elements of your QMS framework. Batch records, Protocols (Validation, Qualification, Stability, etc).Get your QUOTE HERE
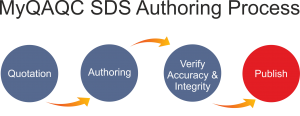
Authoring of MSDS/SDS’ will be done in four simple steps:
- Get a Free Quotation or proposal from us.
While designing your proposal the following is borne in mind
- Identifying your exact compliance needs
- What format MSDS do you need?
- What information do you have about your products?
- When do you need the MSDS?

- Authoring the MSDS
Once we’ve agreed on the deliverables and deadline, our compliance expert will author your MSDS to fulfill your compliance needs.
- Verify Accuracy and Integrity
Once authored, the document is carefully combed through to verify its accuracy and technical integrity. This is done through expert copyediting processes.
- Completing the MSDS
You will be notified when your MSDS is ready to be picked and your project will be released online for your review . Your MSDS will be made available for download in your section on this website.
Technical Benefits
- Classification of substances and/or products to determine the need for labeling and documentation required
- Review of your current SDS against REACH and GHS requirements
- Identification of gaps and required changes
- Conversion of gaps and required changes
- Translation of hazard symbols to GHS standard
- Gap closure for missing data fields
Formats supported:
- REACH Regulation – Registration, Evaluation, Authorisation, and Restriction of Chemical Substances
- CLP Regulation – Regulation on classification, labeling, and packaging of substances and mixtures
- S. Occupational Safety & Health Administration’s (OSHA)
- Hazard Communication Standard
- GHS – Globally Harmonized System of Classification and labeling of Chemicals
- JIS Z7250 – Japanese GHS SDS Standard
